Double Slit Experiment
Christian Huygens, who was a contemporary of Isaac Newton, suggested that light travels in waves. Isaac Newton, however, thought that light was compsed of particles that were too small to detect individually. In 1801 a physicist in England, Thomas Young, performed an experiment that showed that light behaves as a wave. He passed a beam of light through two thin, parallel slits. Alternating bright and dark bands appeared on a white screen some distance from the slit. Young reasoned that if light were made of particles as Newton suggested, only two bright bands of light would be projected on the white surface. The bright and dark bands demonstrated that the slits were causing light waves to interfere with each other. Sometimes this interference is constructive, and the light waves add together to create a bright patch. Sometimes the intereference is destructive and results in the light waves cancelling each other out creating dark patches on the screen.
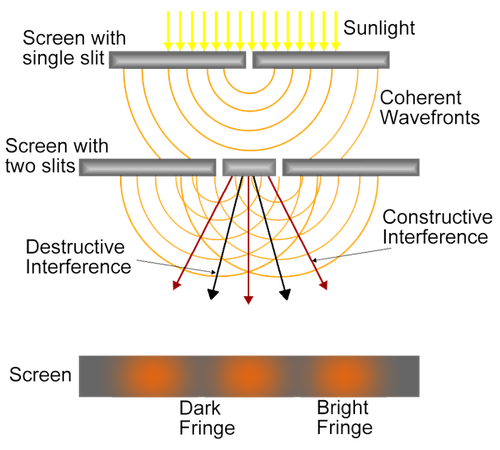
Thomas Young's Double Slit Experiment. Image credit: LCO
Electromagnetic Waves
Electromagnetic waves, including visible light, are made up of oscillating electric and magnetic fields as shown.
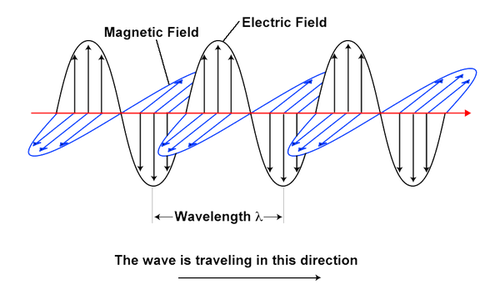
Diagram of electromagnetic waves
The wavelength of a wave is the distance between successive peaks or troughs of a wave. The frequency of a wave is its rate of oscillation and is measured in 1/s. The wavelength of each wave is related to its frequency by the equation λ = c/f where c is the speed of light, f is the frequency of the wave and λ is the wavelength.
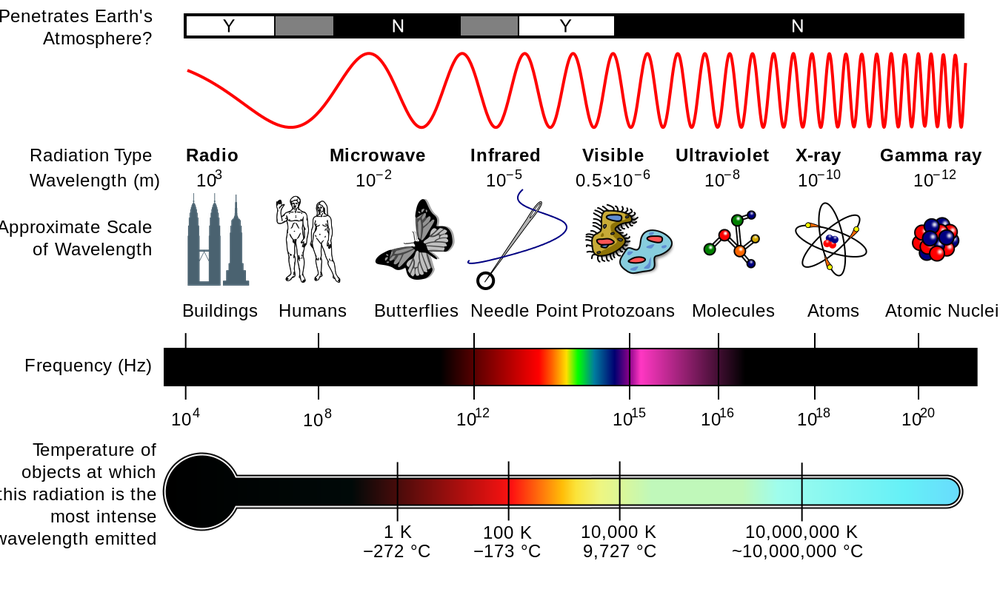
The wavelength of electromagnetic radiation determines the color. All visible light has wavelengths between 400 and 700 nm. Electromagnetic radiation with shorter or longer wavelengths is not visible to humans, but it exists and can be detected. Wavelengths and what type of radiation they correspond to are shown in the table below: